Science & Technology
- Home
- Science & Pipeline
- Science & Technology

Our Technology
Unleashing the Power of Gene Therapy and Nano-antibodies
Our technology represents a paradigm shift in tackling some of humanity's most challenging diseases. By harnessing the combined power of gene therapy and nano-antibodies, we offer targeted solutions where traditional options fall short:
- For children battling rare, debilitating conditions: Precise gene therapy interventions with lipid nano-particles for delivery repair faulty genes at the source, offering hope for a healthier future.
- For individuals struggling with complex neurological disorders: Targeted vectorized nano-antibodies combined with gene therapy deliver powerful cutting-edge treatments directly to affected brain regions, potentially slowing or even reversing disease progression.
- For those facing aggressive cancers: Gene therapy empowers immune cells to recognize and eliminate tumors while specially designed antibody-drug conjugates deliver potent payloads straight to cancer cells which are traditionally resistant to current approaches.
Partnering with the best scientists and clinicians, we are committed to developing effective, personalized treatments that offer renewed hope to patients and their families.
Limitations of Traditional Gene Therapy Approaches:
- Potential immunogenicity
- Limited tissue specificity
- Small loading capacity
- Complex manufacturing
- Cost
Our Advantage
Our proprietary novel next-gen non-viral vectors as delivery vehicles for vectorized nano-antibody gene therapy has many advantages:
-
Biocompatibility exhibits low immunogenicity, reducing the risk of immune responses.
-
Controlled Release Kinetics can be engineered to release their therapeutic cargo in a controlled manner, allowing for sustained gene expression over an extended period.
-
Tuneable Tissue Specificity can be functionalized with various ligands or targeting moieties to enhance their delivery to specific brain regions or cell types.
-
High Capacity have a large surface area and pore volume, allowing them to carry a significantly higher payload of therapeutic genes.
-
Blood-Brain Barrier (BBB) Crossing Ability can be engineered to cross the BBB more effectively, enabling direct delivery of therapeutic genes.
Revolutionizing Neurological Disease Treatment with Dual-Combination Gene Therapy
Addressing the Root Cause of Proteinopathies
Neurological disorders often arise from proteinopathies, characterized by the accumulation of misfolded proteins in the brain. These misfolded proteins disrupt cellular processes, leading to neuronal death and the progressive decline in cognitive and motor functions.
Canary’s dual-combination therapy targets the root cause of proteinopathies by employing two distinct yet synergistic mechanisms:
Vectorized Antibodies

One Target
Our proprietary vectorized antibodies are designed to specifically bind and neutralize misfolded proteins, preventing their aggregation and toxicity. These antibodies are engineered to cross the blood-brain barrier, ensuring effective delivery to the affected areas of the brain.
Functional Transgene Replacement

Multiple Target
We utilize gene therapy to replace defective genes with functional copies, correcting the genetic abnormalities that underlie protein misfolding. This approach restores the production of normal proteins, halting the progression of disease and promoting cellular repair.
The Power of LNP mRNA Precision Medicine
Leveraging Proven Technology to Transform Gene Therapy for Rare Diseases & CNS Disorders
What is LNP & how does it work:
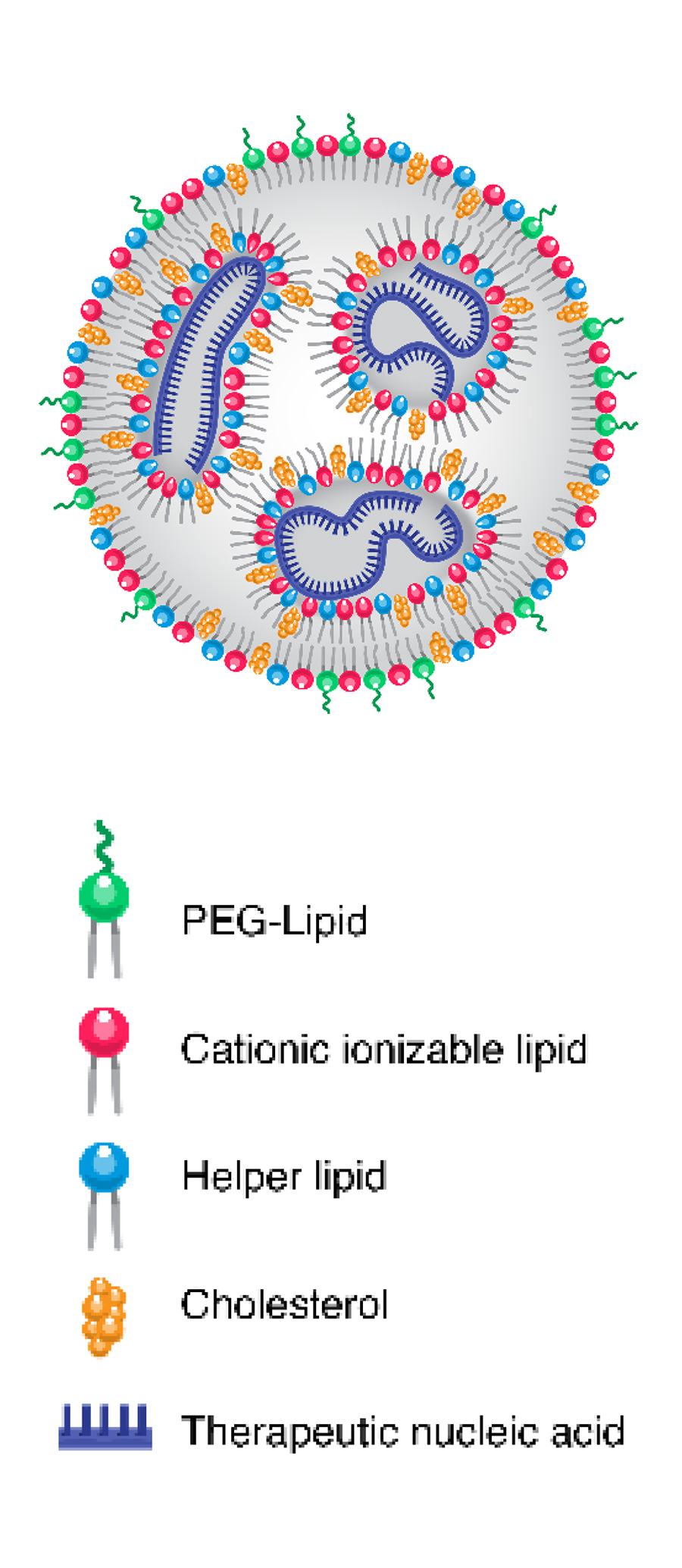
Lipid nanoparticles (LNPs) are delivery vehicles composed of biocompatible lipids (fats) that encapsulate therapeutic agents. They offer important therapy advantages:
- Biocompatibility: Their lipid composition closely resembles biological membranes, minimizing immune system reactions and toxicity.
- Versatility: They can be customized to encapsulate various types of therapeutic agents for diverse applications.
- Targeted Delivery: They can be functionalized with targeting ligands to specifically bind to receptors on cells in the central nervous system (CNS), enhancing drug delivery and reducing side effects.
- Controlled Release: They can be designed to release their cargo gradually over time, providing sustained therapeutic effects.
What is mRNA & how does it work:
- mRNA (messenger ribonucleic acid) is a molecule that carries instructions from DNA in the nucleus to ribosomes in the cytoplasm, telling them which proteins to build.
- The mRNA is encased in lipid particles that protect it and help it enter cells. Once inside the targeted cell, the mRNA instructs the cell's machinery to make copies of specific proteins.
- mRNA was first used in COVID-19 vaccines with great success.
RNAi drug development complemented by AI guided novel biomarker discovery and diagnostics.
Limitations and solutions
The Science of RNAi
Leveraging the body’s naturally occurring ribonucleic acid interference (RNAi) response, Canary is working to develop therapies that target key disease proteins.
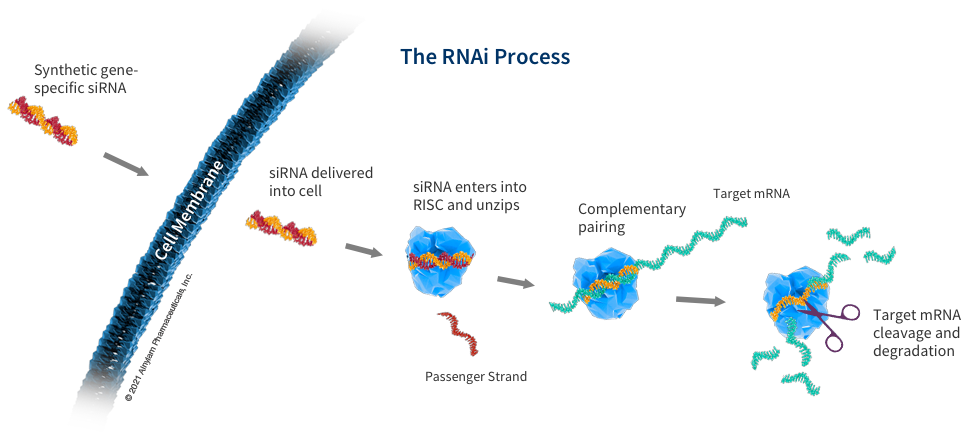
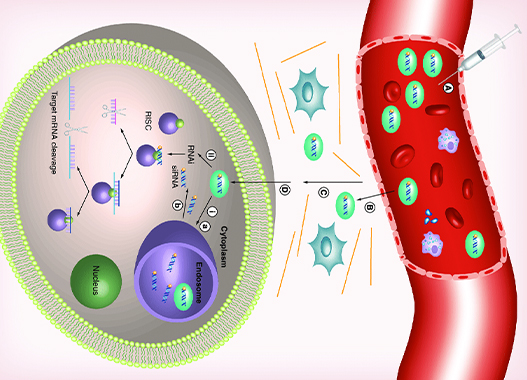
RNAi therapies utilize two short segments of RNA, known as short interfering, or silencing RNA (siRNA), to defeat proteins involved in the progression of certain diseases. The siRNA segments are encapsulated within a histidine-lysine polypeptide (HKP) that ensures their intact delivery to target cells within the body. Once taken up at a cellular level, the siRNA strands are released to activate an enzymatic process driven by the RNA-Induced Silencing Complex (RISC). Through this process, the messenger RNA strands of the disease proteins are targeted and cleaved, ultimately preventing their function to express proteins within a cell.
In addition to the synergistic effect of dual gene/protein targeting, RNAi therapeutics work to prevent the reproduction of harmful proteins within the body, rather than neutralizing existing disease. Additionally, this new class of drugs offers speedier development timelines compared to traditional methods of development involving the screening of small molecules or generating antibodies against a protein. This speedier development along with the potential for earlier treatment offers hope for improving the lives of patients who have been diagnosed with conditions ranging from cancer to fibrotic diseases.
Canary DigiGENE miRNA Diagnostic overview
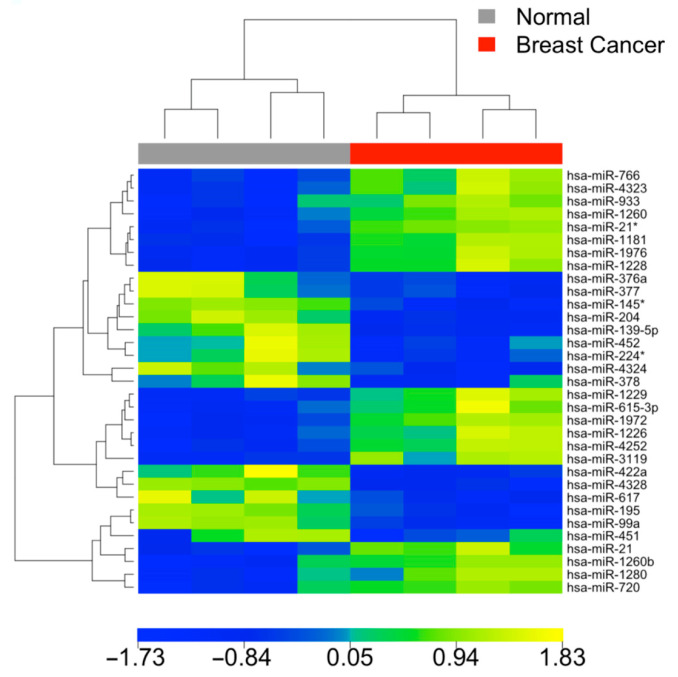
Our point of care diagnostic platform uses Al to read miRNA expression patterns in complex diseases at the earliest stages.
Studies have shown a clear distinction in miRNA expression patterns between disease sample and normal healthy samples. This is a heat map of four breast cancer patients showing separation of miRNA expression profiles in tumors and normal tissues.
WHAT IS THE SCIENCE BEHIND RNA interference?
RNA interference (RNAi) is a natural process that cells use to regulate gene expression. It works by silencing the expression of genes that are involved in disease.
How does RNAi work?
RNAi is a two-step process. In the first step, a double-stranded RNA (dsRNA) molecule is cleaved by an enzyme called Dicer. This produces two single-stranded RNA (ssRNA) molecules, which are called siRNAs (short interfering RNAs).
In the second step, the siRNAs bind to a protein complex called the RNA-induced silencing complex (RISC). The RISC then binds to the target mRNA and cleaves it, preventing it from being translated into protein.
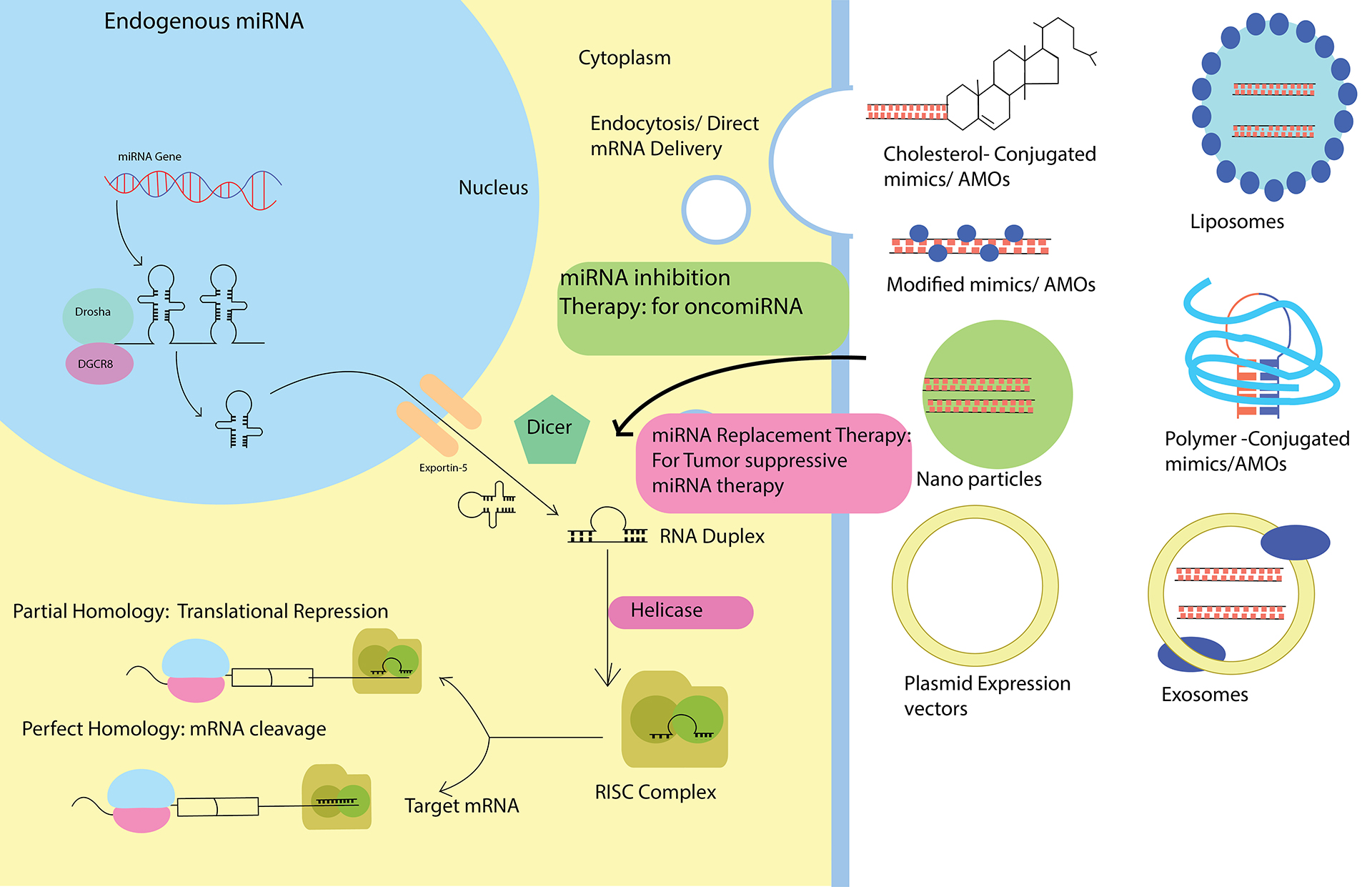
AntiMirs
AntiMirs are short RNA molecules that are designed to bind to and inhibit the function of miRNAs. They can be used to treat diseases that are caused by the overexpression of miRNAs.
AntiMirs can be delivered to cells in a variety of ways, including
- Intravenous injectionThis is the most common method of delivering AntiMirs
- Intramuscular injection This method is used when the target cells are located in the muscle tissue
- Intranasal delivery This method is used to deliver AntiMirs to the cells in the nose and throat
- Inhalation This method is used to deliver AntiMirs to the lungs
- Topical delivery This method is used to deliver AntiMirs to the skin
Canary's AlloMir gene-silencing platform can be leveraged to investigate a variety of diseases and conditions
Advantages of AntiMir technology
Stability
AntiMirs are more stable than siRNAs. This is because they are single-stranded RNA molecules, while siRNAs are double-stranded RNA molecules. Double-stranded RNA molecules are more easily degraded by enzymes in the bloodstream and tissues.
Specificity
AntiMirs can be designed to be more specific than siRNAs. This is because they can be designed to bind to a specific miRNA, while siRNAs can bind to a range of miRNAs.
Safety
AntiMirs may be safer than siRNAs. This is because they are less likely to trigger an immune response.
AntimicroRNA (AntiMir) therapeutics have several potential advantages over siRNA therapeutics:

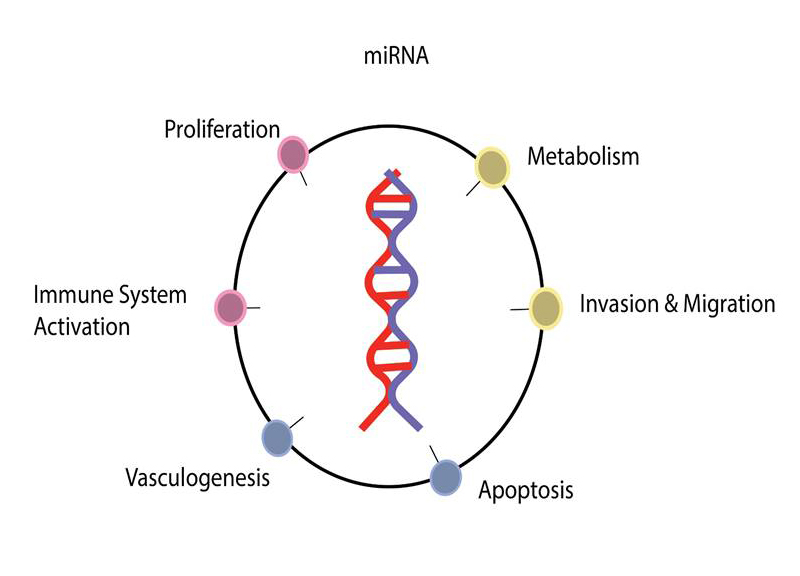
- MicroRNAs (miRNAs) are small RNA molecules that can modulate gene expression to affect numerous biological processes
- microRNAs play a critical role in human biology
- microRNA is important for drug development